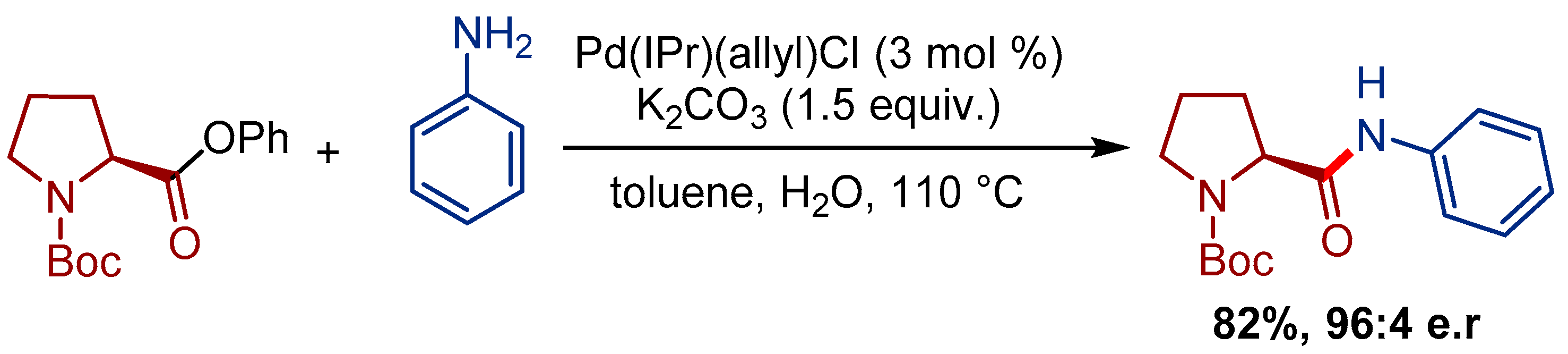
Palladium(ii) allyl complexes of chiral diphosphazane ligands: ambident coordination behaviour and stereodynamic studies in solution - Dalton Transactions (RSC Publishing)
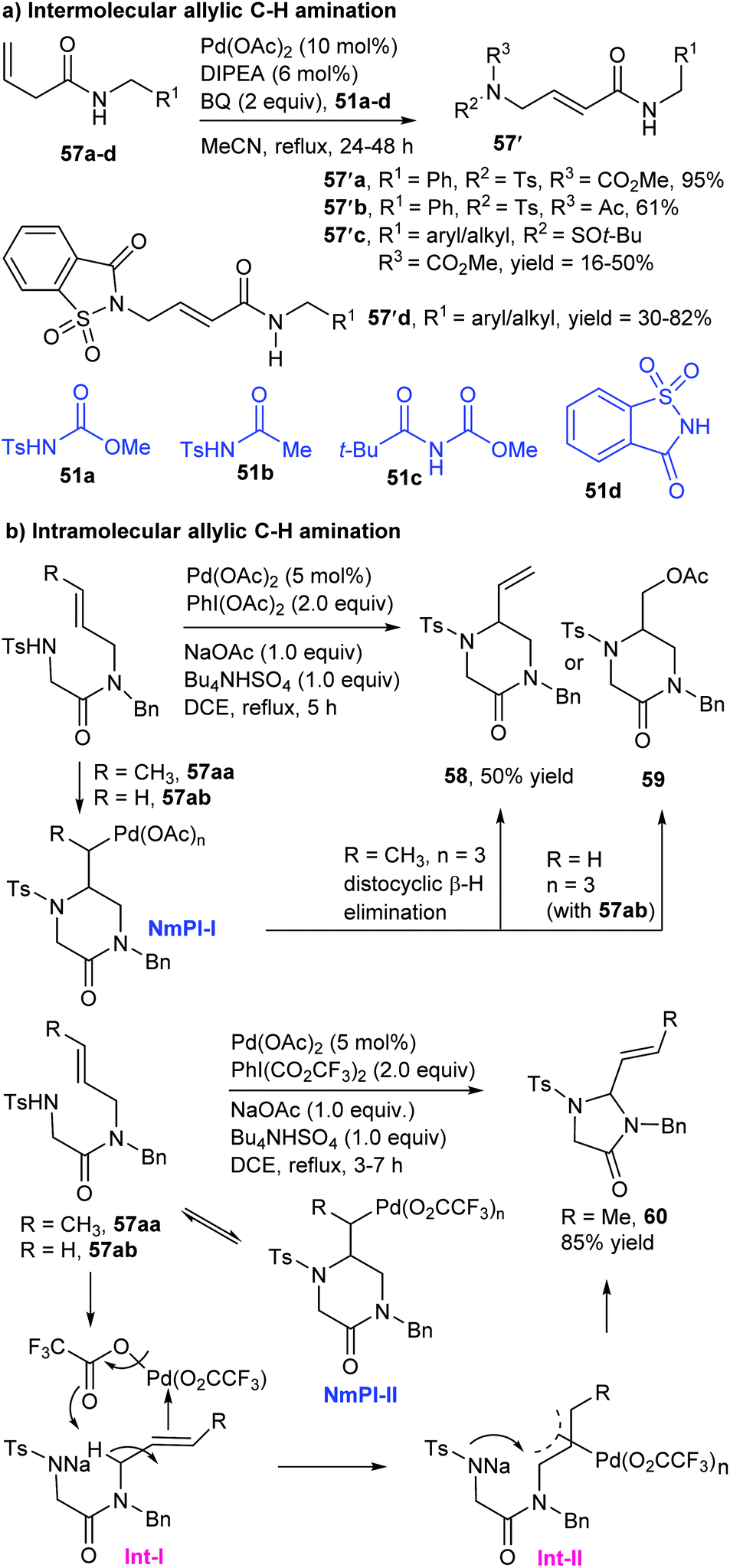
Catalytic allylic functionalization via π-allyl palladium chemistry - Organic & Biomolecular Chemistry (RSC Publishing)

Palladium‐Catalyzed Allylic Substitution: Reversible Formation of Allyl‐Bridged Dinuclear Palladium(I) Complexes - Markert - 2007 - Angewandte Chemie International Edition - Wiley Online Library
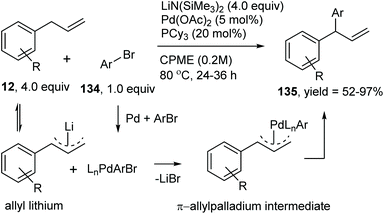
Catalytic allylic functionalization via π-allyl palladium chemistry - Organic & Biomolecular Chemistry (RSC Publishing)
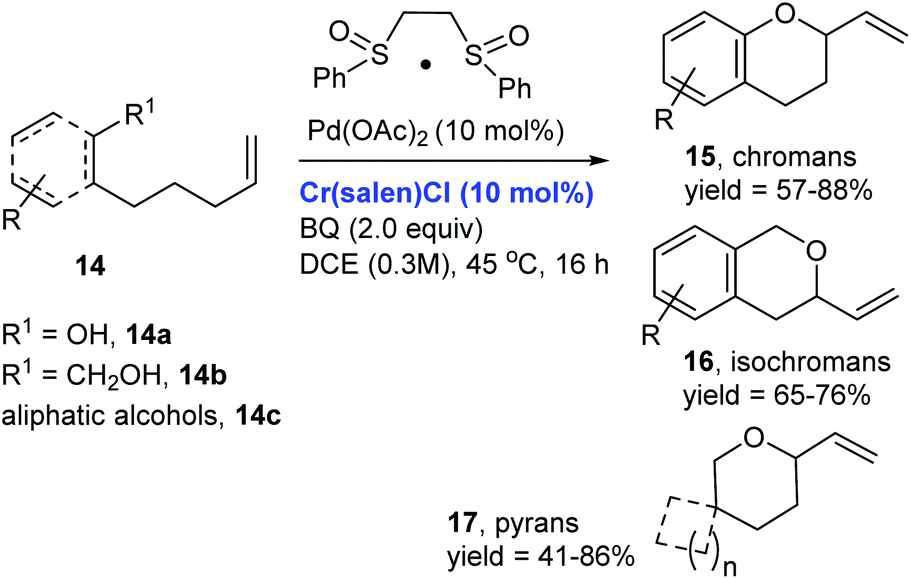
Catalytic allylic functionalization via π-allyl palladium chemistry - Organic & Biomolecular Chemistry (RSC Publishing)

Palladium‐Catalyzed Allylic Substitution at Four‐Membered‐Ring Systems: Formation of η1‐Allyl Complexes and Electrocyclic Ring Opening - Audisio - 2013 - Angewandte Chemie International Edition - Wiley Online Library

Palladium‐Catalyzed Electrophilic Allylation Reactions via Bis(allyl) palladium Complexes and Related Intermediates - Szabó - 2004 - Chemistry – A European Journal - Wiley Online Library

Palladium-catalyzed reaction of γ-silylated allyl acetates proceeding through 1,2-shift of a substituent on silicon - ScienceDirect

Palladium prompted on-demand cysteine chemistry for the synthesis of challenging and uniquely modified proteins | Nature Communications

Schematic representation of palladium-mediated modification of peptide... | Download Scientific Diagram

Reaction rate between allyl alcohol and Pd(TPPTS) 3 : H + /Pd ratio... | Download Scientific Diagram

Palladium TPPTS catalyst in water: C-allylation of phenol and guaiacol with allyl alcohol and novel isomerisation of allyl ethers of phenol and guaiacol - ScienceDirect

Steric Effects in Enantioselective Allylic Alkylation Catalysed by Cationic(η3‐Allyl)palladium Complexes Bearing Chiral Pyridine‐Aziridine Ligands - Ferioli - 2005 - European Journal of Organic Chemistry - Wiley Online Library

Synthesis and comparative study of the anticancer activity of η3-allyl palladium(II) complexes bearing N-heterocyclic carbenes as ancillary ligands - ScienceDirect

Palladium: Organometallic Chemistry - William Suggs - - Major Reference Works - Wiley Online Library

Palladium-catalyzed reaction of γ-silylated allyl acetates proceeding through 1,2-shift of a substituent on silicon - ScienceDirect
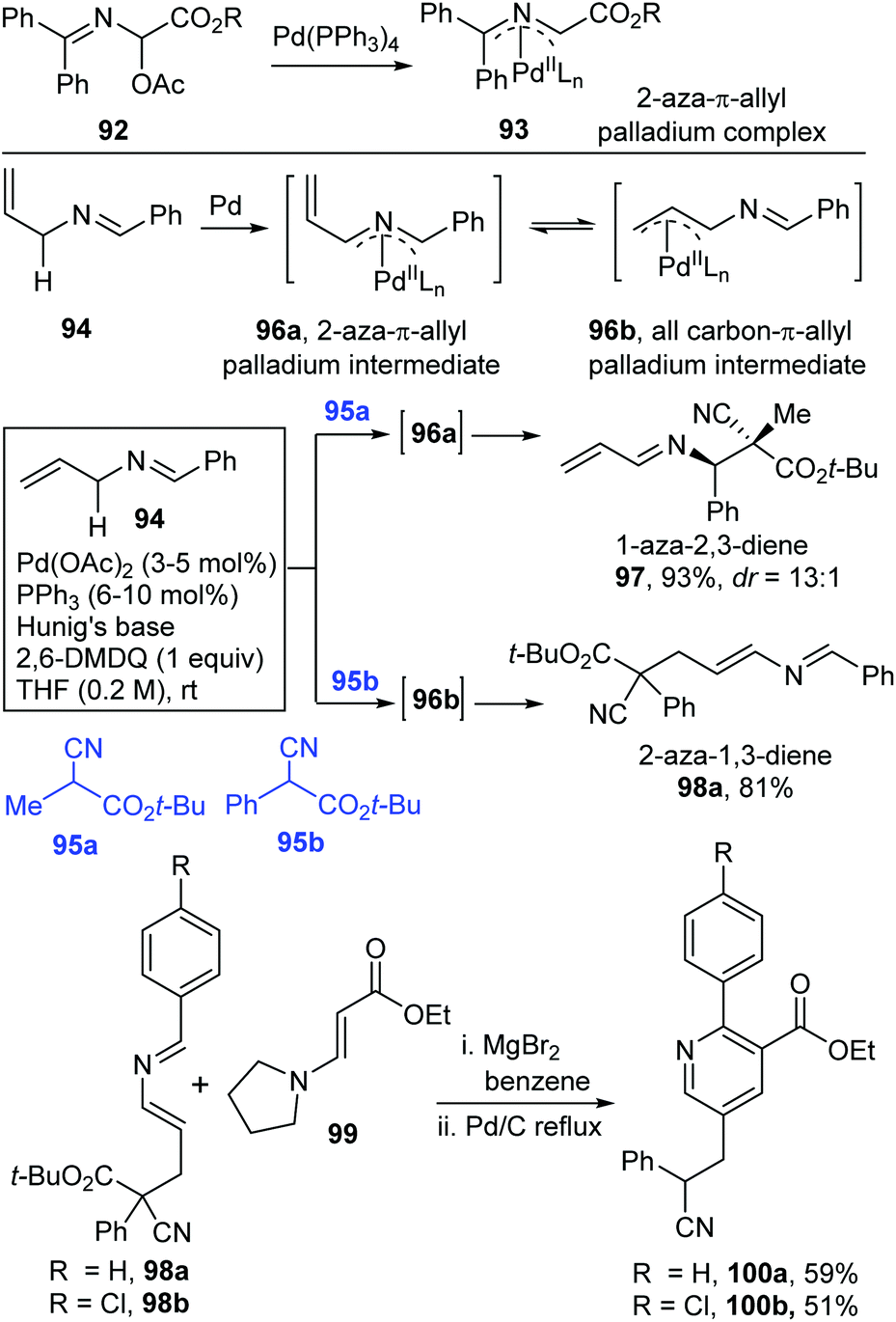
Catalytic allylic functionalization via π-allyl palladium chemistry - Organic & Biomolecular Chemistry (RSC Publishing)

Protonation of palladium(II)-allyl and palladium(0)-olefin complexes containing 2-pyridyldiphenylphosphine - ScienceDirect
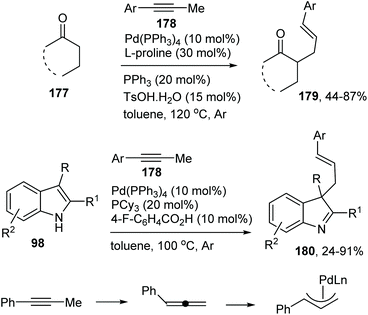
Catalytic allylic functionalization via π-allyl palladium chemistry - Organic & Biomolecular Chemistry (RSC Publishing)
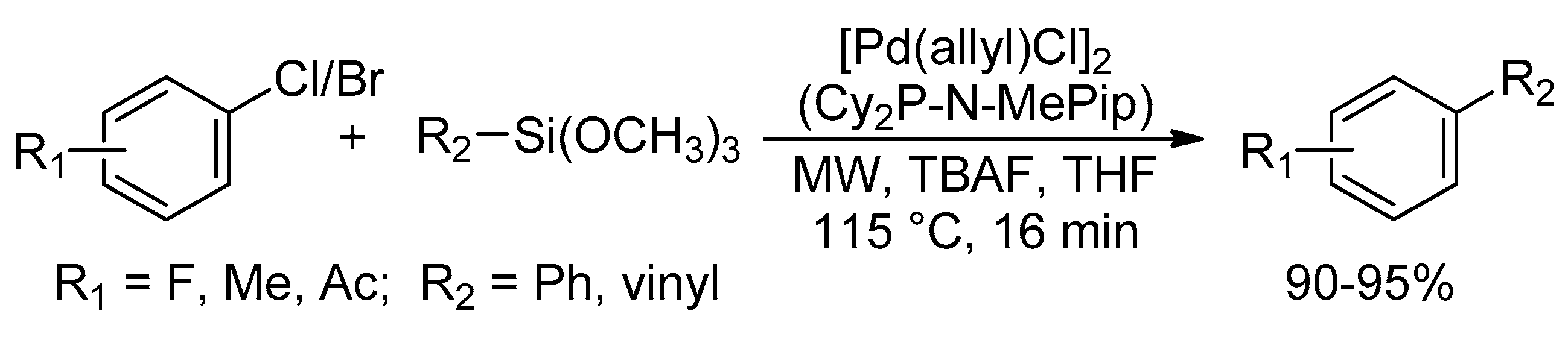
Catalysts | Free Full-Text | Microwave-Assisted Palladium-Catalyzed Cross-Coupling Reactions: Generation of Carbon–Carbon Bond

Palladium‐Catalyzed Electrophilic Allylation Reactions via Bis(allyl) palladium Complexes and Related Intermediates - Szabó - 2004 - Chemistry – A European Journal - Wiley Online Library
A Mechanistic Study of Direct Activation of Allylic Alcohols in Palladium Catalyzed Amination Reactions