Absolute stereochemistry and limitations of the palladium-catalysed... | Download Scientific Diagram

Palladium-catalyzed allylation and deacylative allylation of 3-acetyl-2-oxindoles with allylic alcohols - ScienceDirect

Palladium TPPTS catalyst in water: C-allylation of phenol and guaiacol with allyl alcohol and novel isomerisation of allyl ethers of phenol and guaiacol - ScienceDirect

Palladium-Catalyzed meta-C-H Allylation of Arenes: A Unique Combination of a Pyrimidine-Based Template and Hexafluoroisopropanol. - J. Am. Chem. Soc. - X-MOL
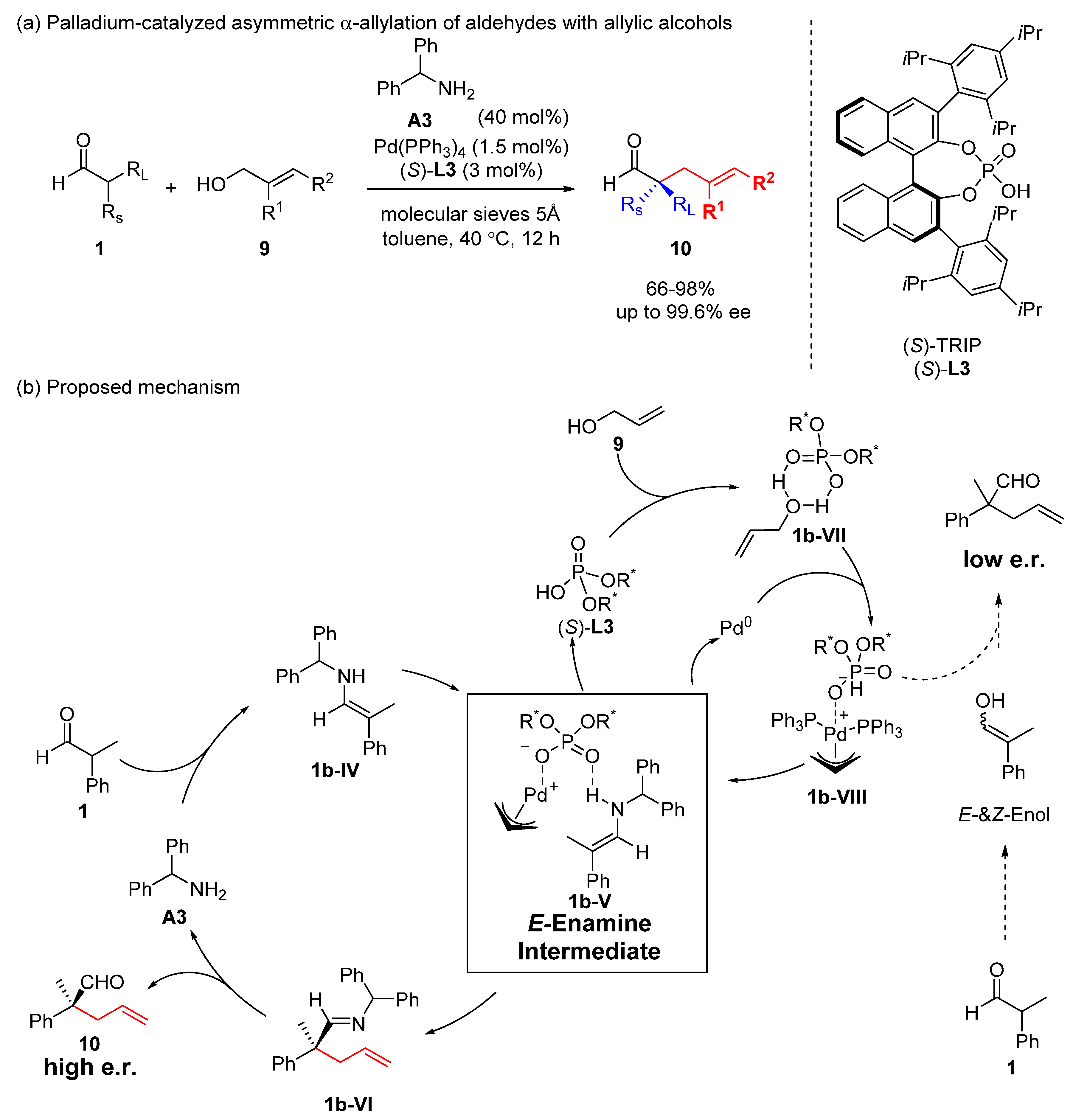
Catalysts | Free Full-Text | Transition Metal-Catalyzed α-Position Carbon–Carbon Bond Formations of Carbonyl Derivatives | HTML
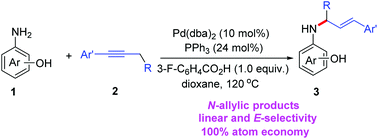
Palladium-catalyzed allylation of aminophenol with alkynes to construct C–N bonds - Organic & Biomolecular Chemistry (RSC Publishing)

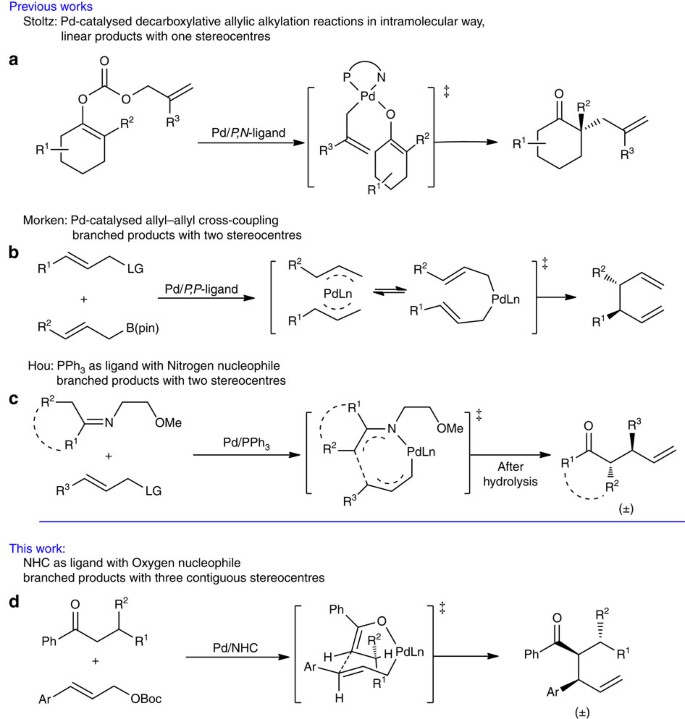
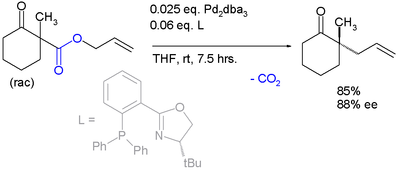
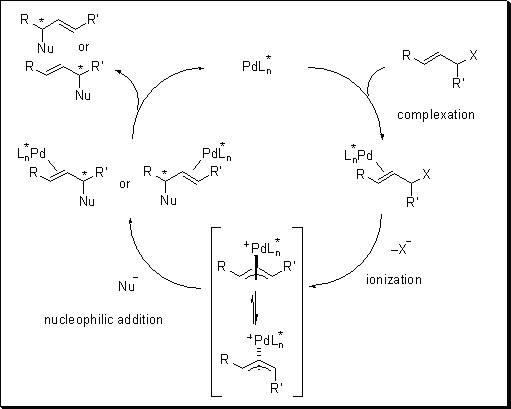
Palladium‐Catalyzed Decarboxylative Asymmetric Allylic Alkylation: Development, Mechanistic Understanding and Recent Advances - James - 2019 - Advanced Synthesis & Catalysis - Wiley Online Library

EurJOC on Twitter: "Palladium-Catalyzed Asymmetric Decarboxylative Allylation of Azlactone Enol Carbonates: Fast Access to Enantioenriched alpha-Allyl Quaternary Amino Acids (Colombo) https://t.co/hnX2TG6PxH… https://t.co/VOtquqwWIy"

Palladium‐Catalyzed Decarboxylative Asymmetric Allylic Alkylation: Development, Mechanistic Understanding and Recent Advances - James - 2019 - Advanced Synthesis & Catalysis - Wiley Online Library
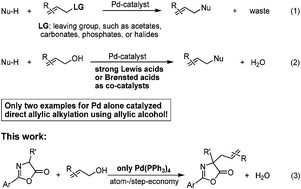
Palladium catalyzed direct allylation of azlactones with simple allylic alcohols in the absence of any activators - RSC Advances (RSC Publishing)
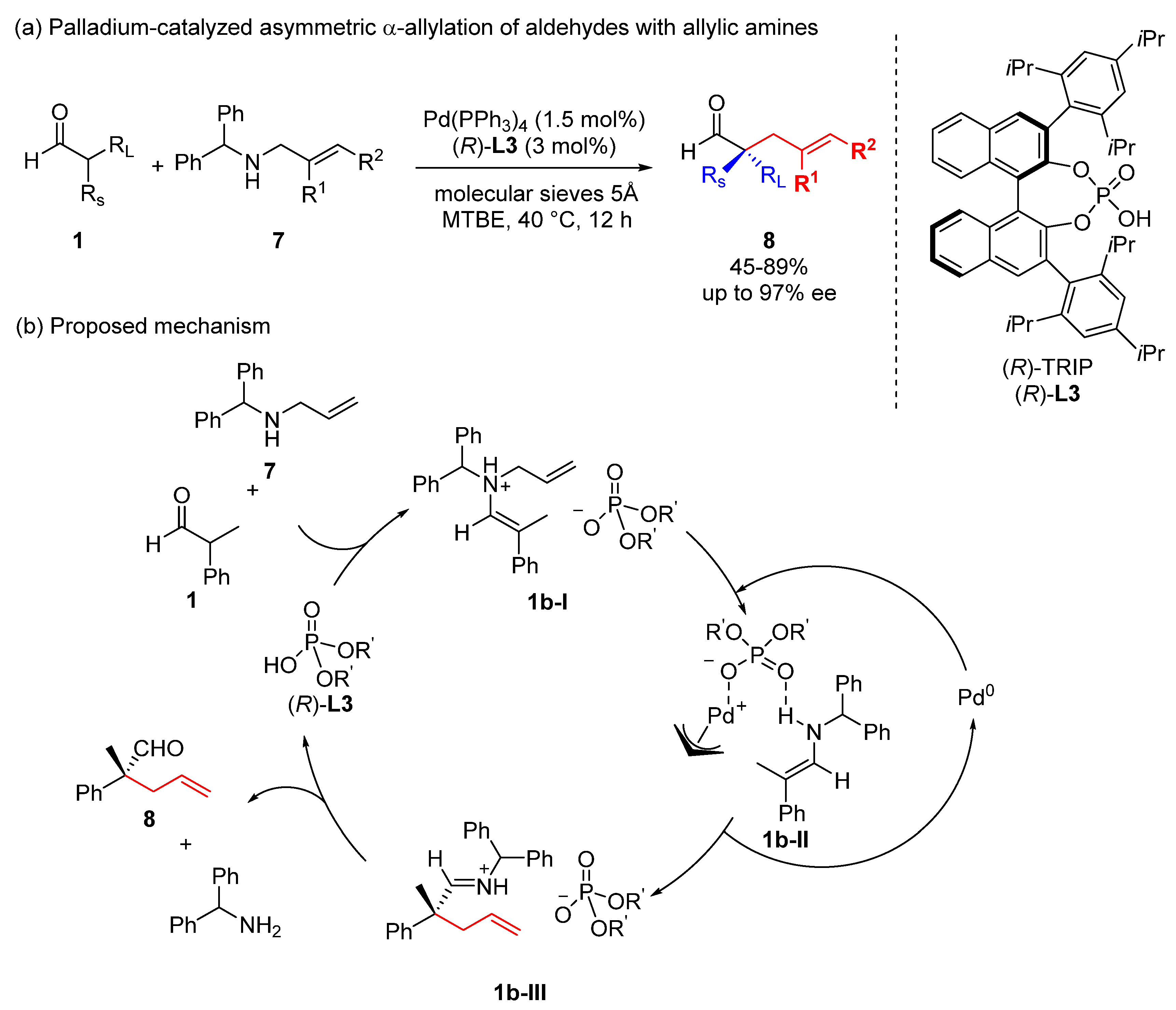
Catalysts | Free Full-Text | Transition Metal-Catalyzed α-Position Carbon–Carbon Bond Formations of Carbonyl Derivatives | HTML
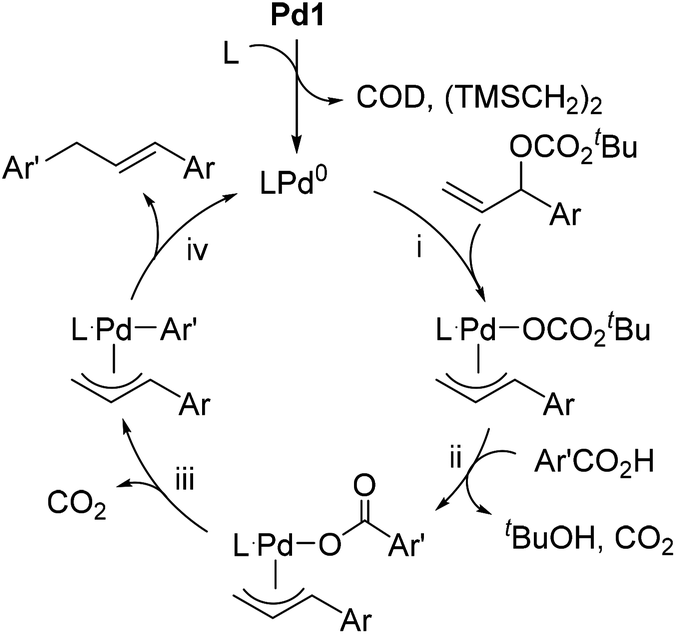
Palladium-catalyzed salt-free double decarboxylative aryl allylation - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C8OB01806E
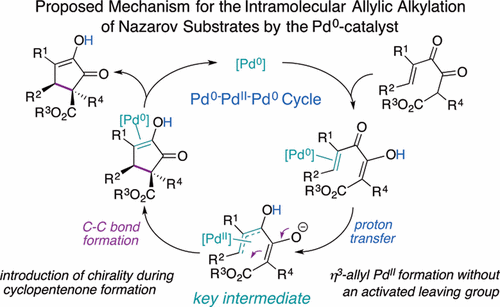
It Is Not Just Up to the Substrate: Palladium(0) Cyclizes Nazarov Substrates through Intramolecular Allylic Alkylation - Organometallics - X-MOL

Palladium-Catalyzed Synthesis of Substituted Cycloheptane-1,4-diones by an Asymmetric Ring-Expanding Allylation (AREA)
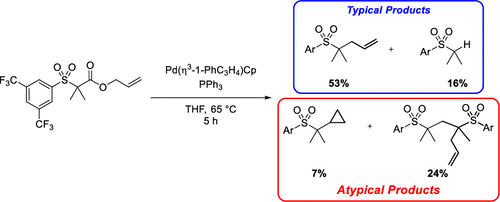
Development of Palladium-Catalyzed Decarboxylative Allylation of Electron-Deficient Sulfones and Identification of Unusual Side Products - J. Org. Chem. - X-MOL