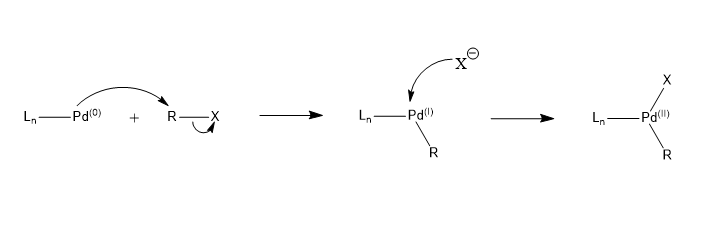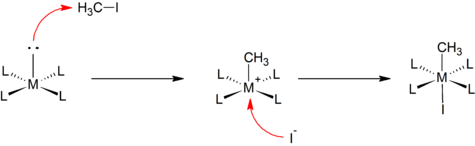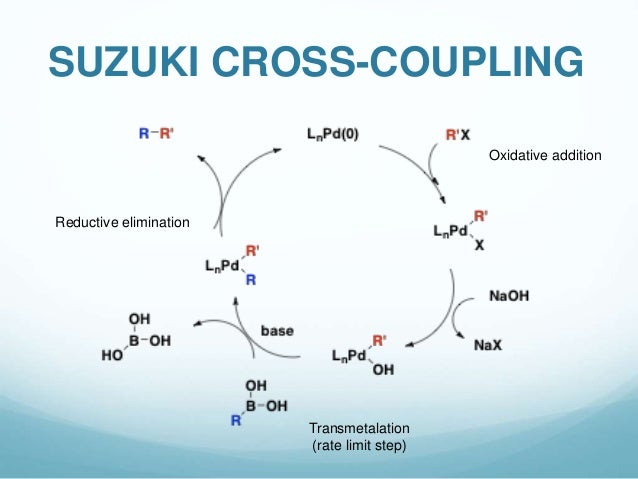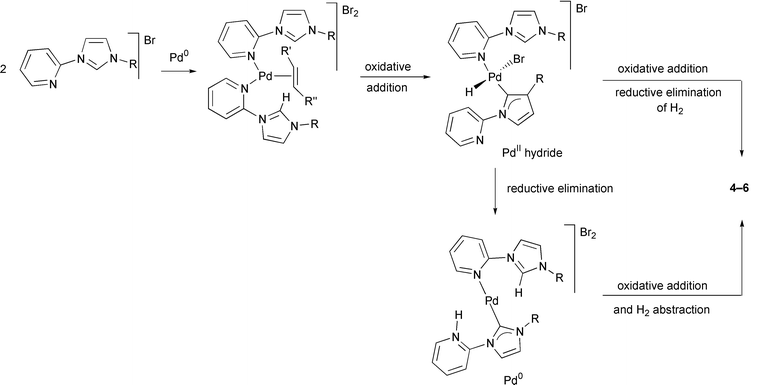
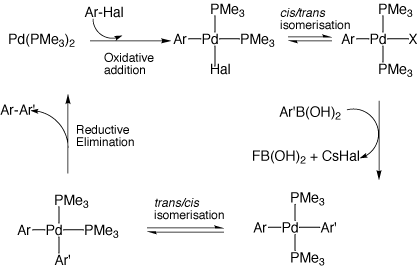
Bis-carbene complexes from oxidative addition of imidazolium C–H bonds to palladium(0) - Journal of the Chemical Society, Dalton Transactions (RSC Publishing)

Formation of XPhos‐Ligated Palladium(0) Complexes and Reactivity in Oxidative Additions - Wagschal - 2019 - Chemistry – A European Journal - Wiley Online Library

Oxidative addition of organic halides on palladium(0) complexes stabilized by dimethylfumarate and quinoline-based N–P or N–S spectator ligands - ScienceDirect

Report: Toward Greater Understanding and Expanded Utility of the Palladium-Catalyzed Activation of Carbon-Carbon Single Bonds (56th Annual Report on Research Under Sponsorship of The American Chemical Society Petroleum Research Fund)
A Computational Study of the Mechanism of Palladium Insertion into Alkynyl and Aryl Carbon-Fluorine bonds
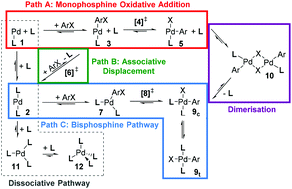
Computed ligand effects on the oxidative addition of phenyl halides to phosphine supported palladium(0) catalysts - Dalton Transactions (RSC Publishing)
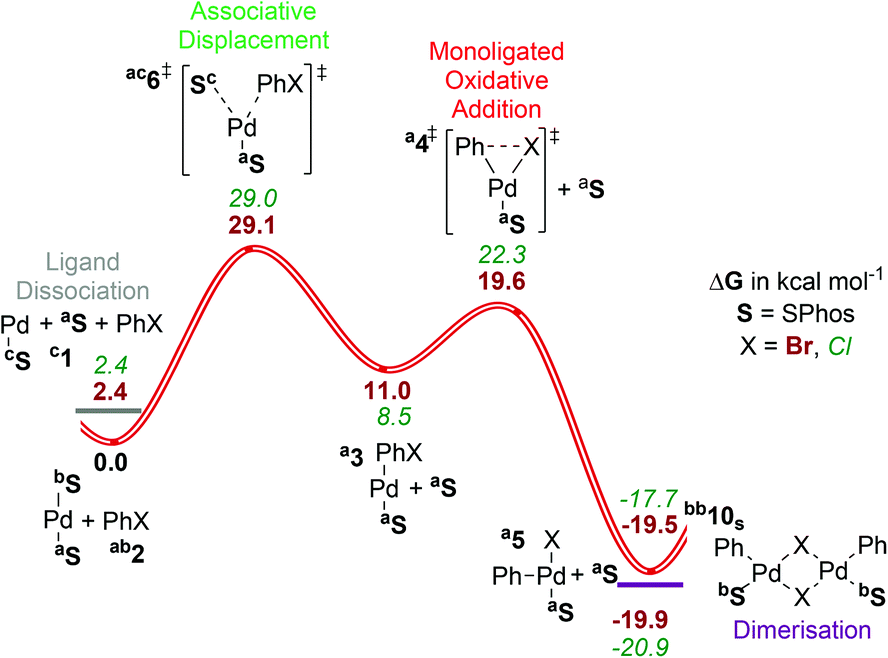
Computed ligand effects on the oxidative addition of phenyl halides to phosphine supported palladium(0) catalysts - Dalton Transactions (RSC Publishing) DOI:10.1039/C4DT01758G
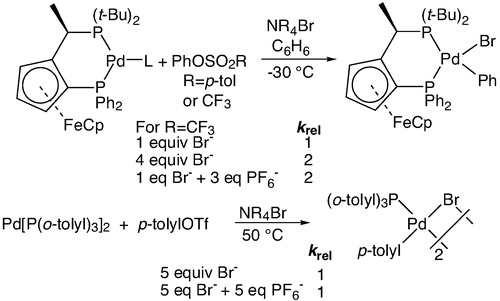
Oxidative Addition of Aryl Sulfonates to Palladium(0) Complexes of Mono- and Bidentate Phosphines. Mild Addition of Aryl Tosylates and the Effects of Anions on Rate and Mechanism | The Hartwig Group
Interpreting Oxidative Addition of Ph–X (X = CH3, F, Cl, and Br) to Monoligated Pd(0) Catalysts Using Molecular Electrostatic
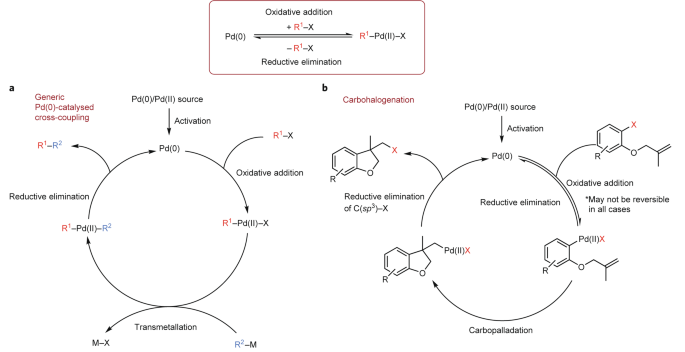
The emergence of Pd-mediated reversible oxidative addition in cross coupling, carbohalogenation and carbonylation reactions | Nature Catalysis
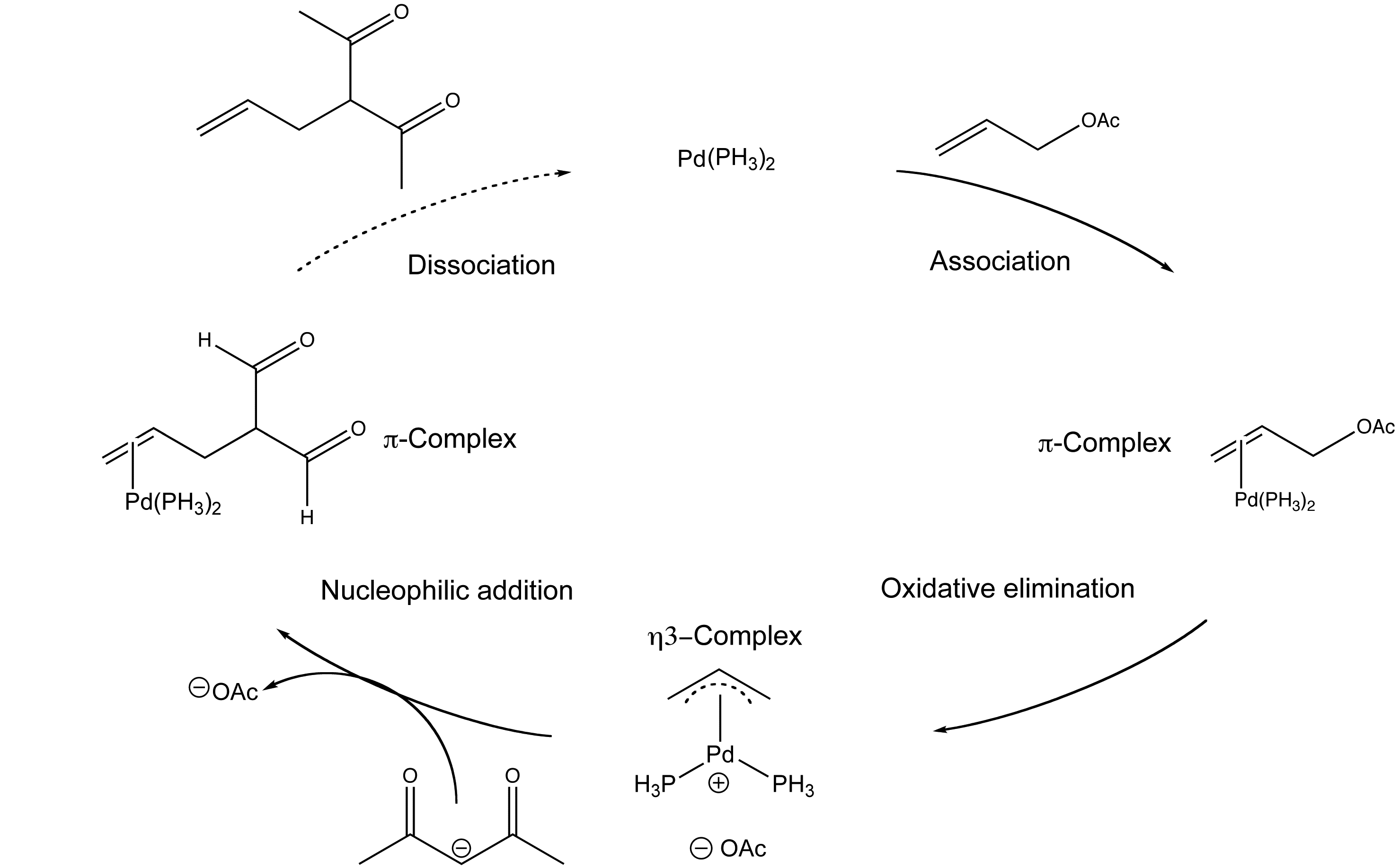
Organopalladium Chemistry - Palladium-catalysed nucleophilic allylic substitution of functionalised compounds