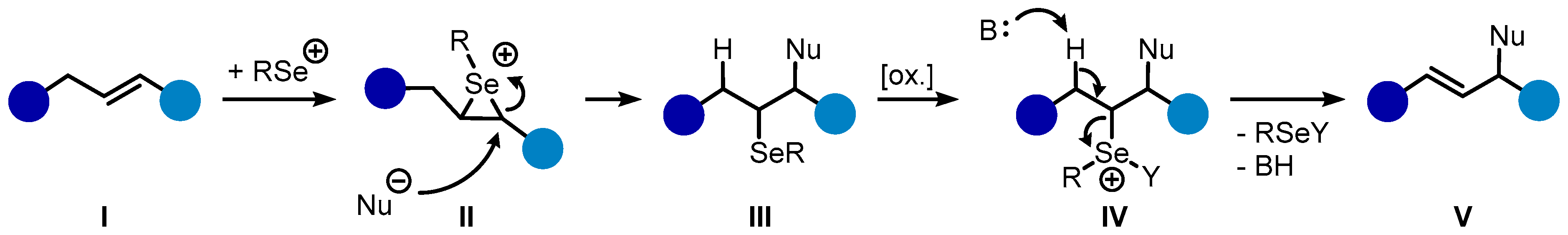
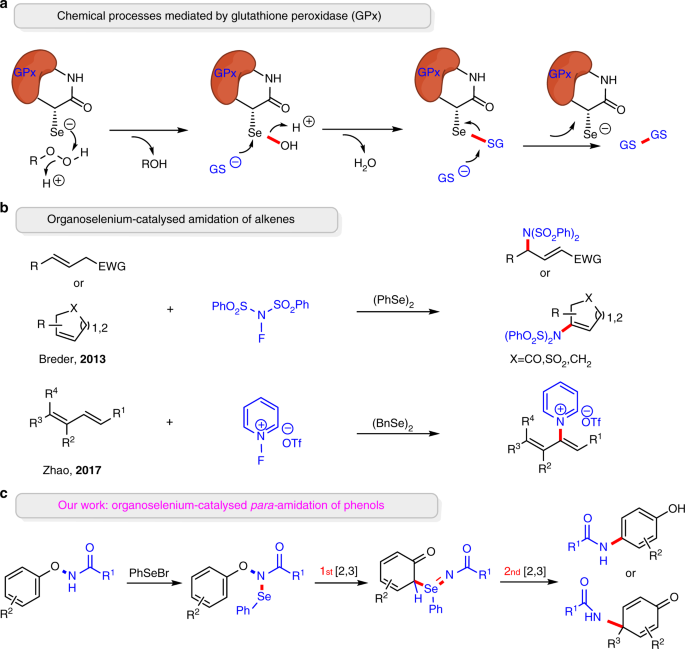
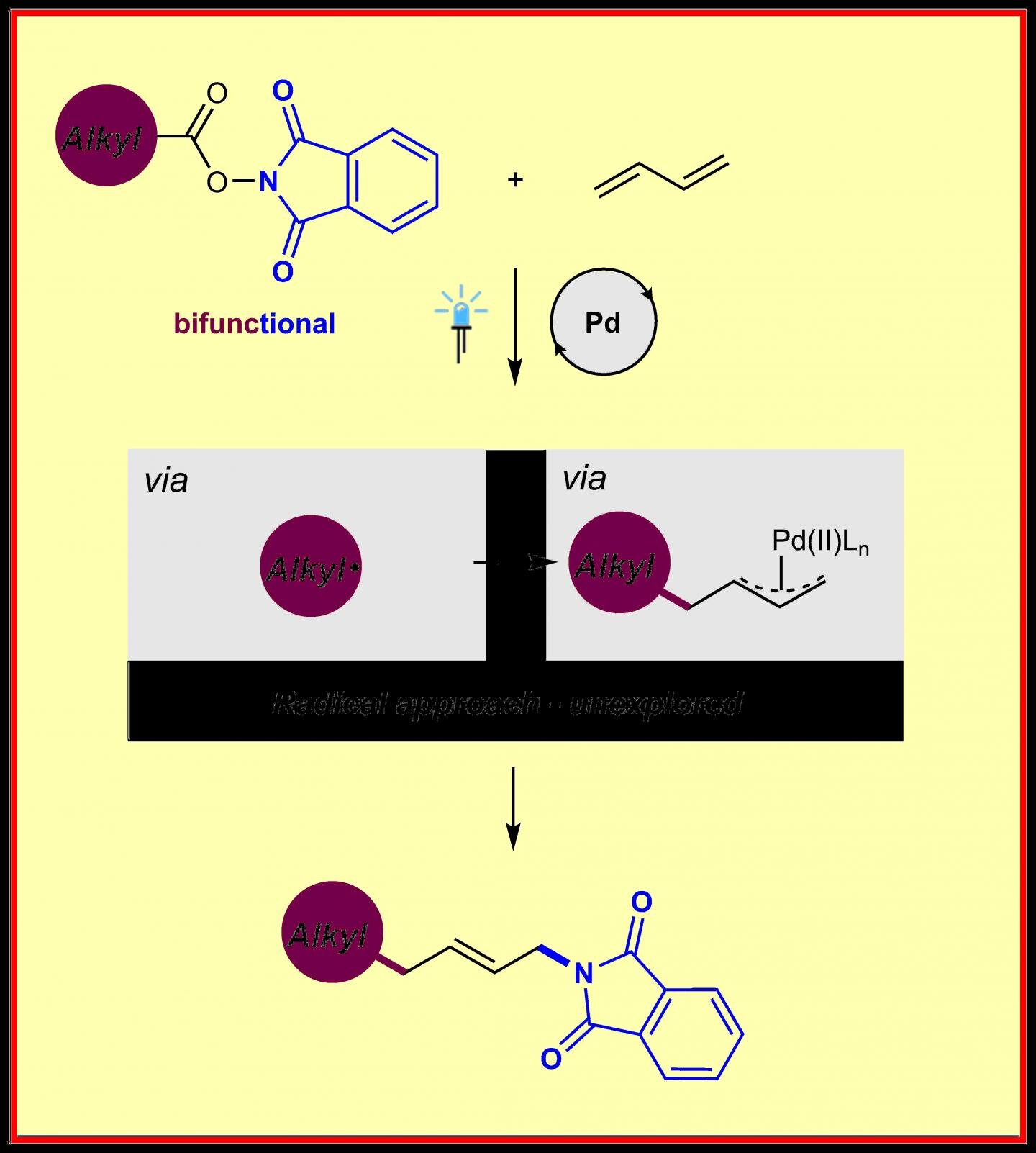
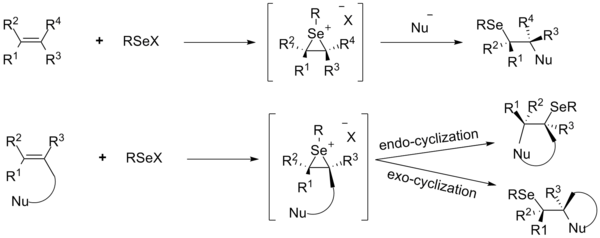
Bis(selone) Complexes of Palladium(II), Platinum(II), and Gold(III): Synthesis and Structural Studies - Rani - 2017 - European Journal of Inorganic Chemistry - Wiley Online Library
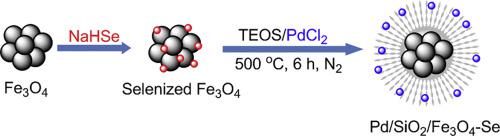
Selenium-directed synthesis of Pd nanoparticles on mesoporous silica-coated Fe3O4: An efficient magnetic catalyst for oxidative alkene cracking - Catal. Commun. - X-MOL
![A selenium-coordinated palladium(ii) trans-dichloride molecular rotor as a catalyst for site-selective annulation of 2-arylimidazo[1,2-a]pyridines - Chemical Communications (RSC Publishing) A selenium-coordinated palladium(ii) trans-dichloride molecular rotor as a catalyst for site-selective annulation of 2-arylimidazo[1,2-a]pyridines - Chemical Communications (RSC Publishing)](https://pubs.rsc.org/en/Content/Image/GA/D0CC03599H)
A selenium-coordinated palladium(ii) trans-dichloride molecular rotor as a catalyst for site-selective annulation of 2-arylimidazo[1,2-a]pyridines - Chemical Communications (RSC Publishing)

The Palladium Slow‐Release Pre‐Catalysts and Nanoparticles in the “Phosphine‐Free” Mizoroki–Heck and Suzuki–Miyaura Reactions - Sigeev - 2015 - Advanced Synthesis & Catalysis - Wiley Online Library
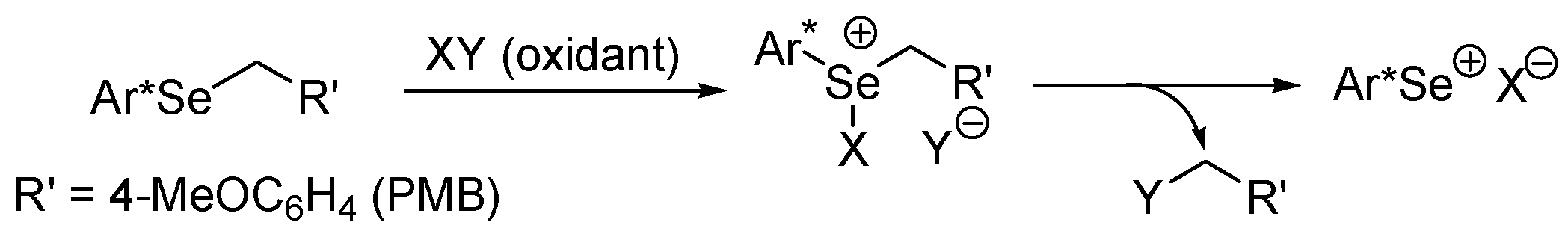
Molecules | Free Full-Text | Electrophilic Selenium Catalysis with Electrophilic N-F Reagents as the Oxidants | HTML
Palladium(ii) complexes bearing the 1,2,3-triazole based organosulfur/ selenium ligand: synthesis, structure and applications in Heck and Suzuki–Miyaura coupling as a catalyst via palladium nanoparticles - RSC Advances (RSC Publishing)

Modular chiral selenium-containing oxazolines: synthesis and application in the palladium-catalyzed asymmetric allylic alkylation - ScienceDirect

A nanoscale lattice of palladium and yttrium makes for a superlative carbon-linking catalyst | EurekAlert! Science News
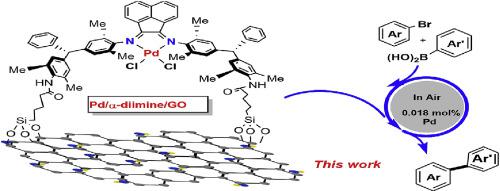
Bulky α-diimine palladium complexes supported graphene oxide as heterogeneous catalysts for Suzuki-Miyaura reaction - J. Mol. Struct. - X-MOL
![A selenium-coordinated palladium(ii) trans-dichloride molecular rotor as a catalyst for site-selective annulation of 2-arylimidazo[1,2-a]pyridines - Chemical Communications (RSC Publishing) A selenium-coordinated palladium(ii) trans-dichloride molecular rotor as a catalyst for site-selective annulation of 2-arylimidazo[1,2-a]pyridines - Chemical Communications (RSC Publishing)](https://pubs.rsc.org/image/article/2020/cc/d0cc03599h/d0cc03599h-s1_hi-res.gif)
A selenium-coordinated palladium(ii) trans-dichloride molecular rotor as a catalyst for site-selective annulation of 2-arylimidazo[1,2-a]pyridines - Chemical Communications (RSC Publishing)
![A selenium-coordinated palladium(ii) trans-dichloride molecular rotor as a catalyst for site-selective annulation of 2-arylimidazo[1,2-a]pyridines - Chemical Communications (RSC Publishing) A selenium-coordinated palladium(ii) trans-dichloride molecular rotor as a catalyst for site-selective annulation of 2-arylimidazo[1,2-a]pyridines - Chemical Communications (RSC Publishing)](https://pubs.rsc.org/image/article/2020/cc/d0cc03599h/d0cc03599h-s2_hi-res.gif)
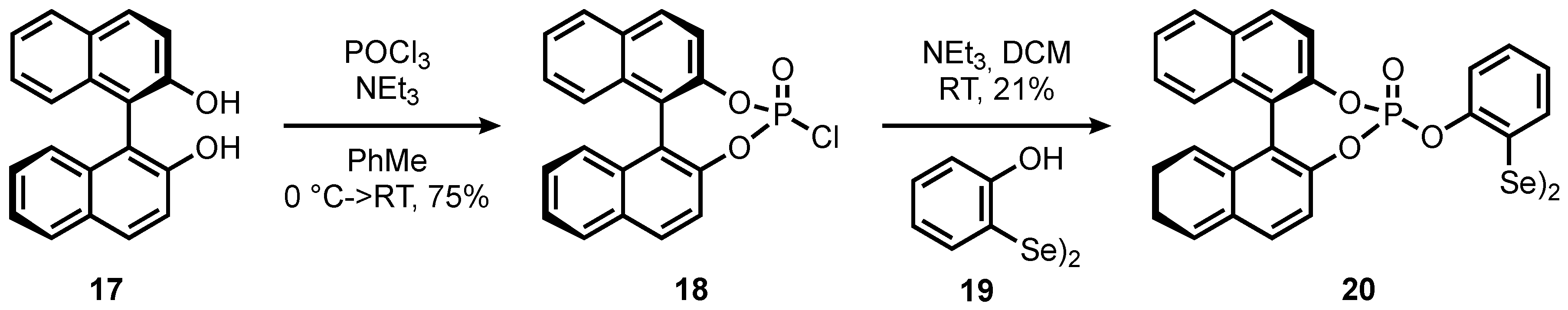
A selenium-coordinated palladium(ii) trans-dichloride molecular rotor as a catalyst for site-selective annulation of 2-arylimidazo[1,2-a]pyridines - Chemical Communications (RSC Publishing)

Table 1 from Selenium-Catalyzed C(sp3)-H Acyloxylation: Application in the Expedient Synthesis of Isobenzofuranones. | Semantic Scholar
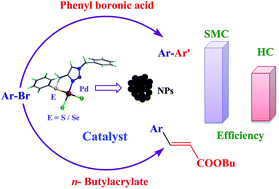
Palladium(ii) complexes bearing the 1,2,3-triazole based organosulfur/ selenium ligand: synthesis, structure and applications in Heck and Suzuki–Miyaura coupling as a catalyst via palladium nanoparticles - RSC Advances (RSC Publishing)

PPT - Selected Catalytic Reactions Involving Selenium Compounds PowerPoint Presentation - ID:3305940
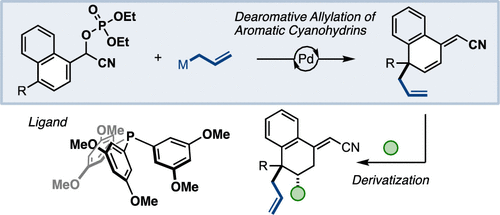
Dearomative Allylation of Naphthyl Cyanohydrins by Palladium Catalysis: Catalyst-Enhanced Site Selectivity. - Org. Lett. - X-MOL

Selenium-catalyzed oxidative carbonylation of 2-aminobenzyl alcohol to give 1,4-dihydro-2 H -3,1-benzoxazin-2-one - ScienceDirect