![PDF) Chlorido-{2-[(di-methyl-amino)-meth-yl]benzene-seleno-lato-κ(2) N,Se}(tri-phenyl-phosphane-κP)palladium(II) PDF) Chlorido-{2-[(di-methyl-amino)-meth-yl]benzene-seleno-lato-κ(2) N,Se}(tri-phenyl-phosphane-κP)palladium(II)](https://i1.rgstatic.net/publication/263208607_Chlorido-2-di-methyl-amino-meth-ylbenzene-seleno-lato-k2_NSetri-phenyl-phosphane-kPpalladiumII/links/550401ab0cf24cee39fdb3ca/largepreview.png)
PDF) Chlorido-{2-[(di-methyl-amino)-meth-yl]benzene-seleno-lato-κ(2) N,Se}(tri-phenyl-phosphane-κP)palladium(II)
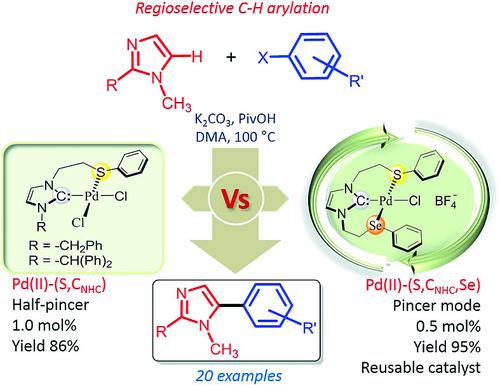
Palladium Complexes of Thio/Seleno‐Ether Containing N‐Heterocyclic Carbenes: Efficient and Reusable Catalyst for Regioselective C‐H Bond Arylation - Eur. J. Inorg. Chem. - X-MOL
![A novel single source precursor: [bis(N,N-diethyl-N′-naphthoyl-selenoureato) palladium(ii)] for palladium selenide thin films and nanoparticles - Chemical Communications (RSC Publishing) A novel single source precursor: [bis(N,N-diethyl-N′-naphthoyl-selenoureato) palladium(ii)] for palladium selenide thin films and nanoparticles - Chemical Communications (RSC Publishing)](https://pubs.rsc.org/en/Content/Image/GA/C0CC03079A)
A novel single source precursor: [bis(N,N-diethyl-N′-naphthoyl-selenoureato) palladium(ii)] for palladium selenide thin films and nanoparticles - Chemical Communications (RSC Publishing)

Palladium-Catalyzed Carbonylation of 2-Haloselenophenes: Synthesis of Selenophene-2-carboxamides, Selenophene-2,5-dicarboxamides and N,N′-Bridged Selenophene-2-carboxamides

Palladium(II) complexes of hemilabile NNS and NNSe iminophosphorane ligands: Synthesis, characterization, and reactivity - ScienceDirect

Applications of metal selenium/tellurium compounds in materials science in: Physical Sciences Reviews Volume 4 Issue 5 (2018)

Palladium complexes of C/Se and N/E (E = Se, Te) ligands, 2-(3,5-dimethylpyrazol-1-yl)phenyl substituted chalcogeno-ethers: Functional group effect on structure and catalytic activity - ScienceDirect

Nanomolar Detection of Palladium (II) through a Novel Seleno-Rhodamine-based fluorescent and colorimetric chemosensor - ScienceDirect
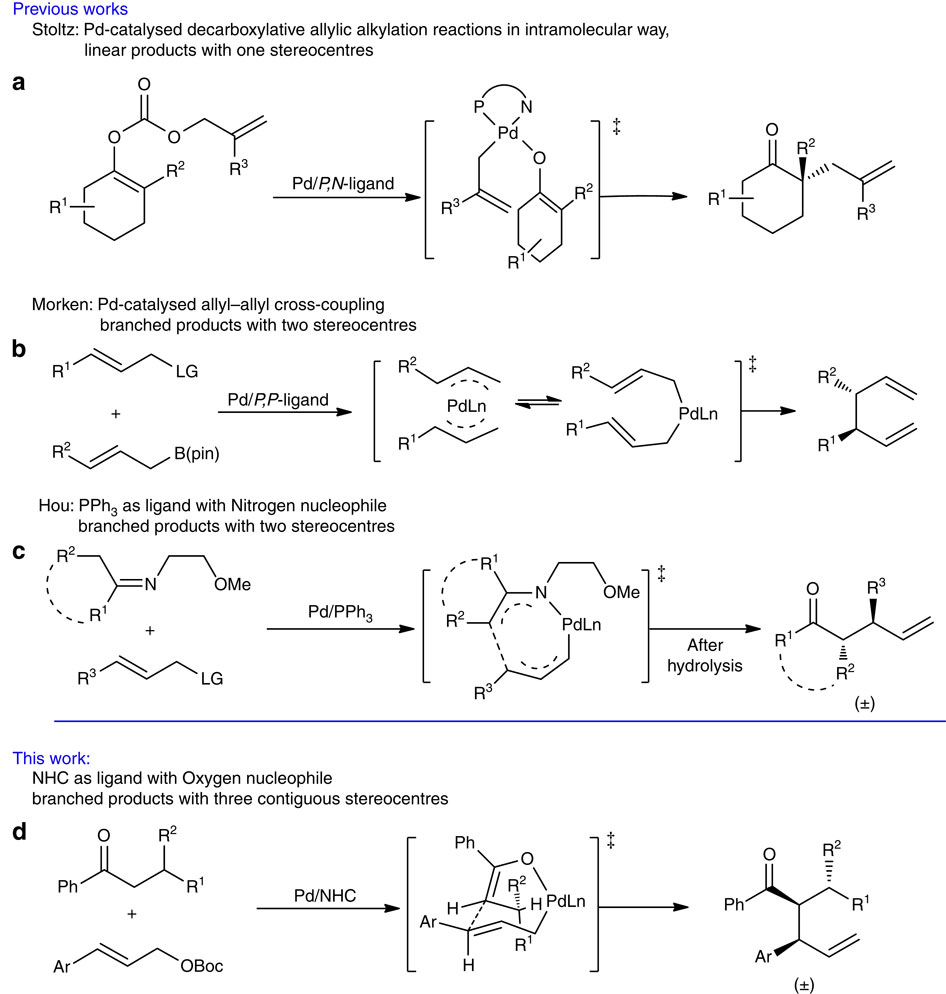
Palladium/N-heterocyclic carbene catalysed regio and diastereoselective reaction of ketones with allyl reagents via inner-sphere mechanism. - Nat. Commun. - X-MOL

Microwave‐Mediated Palladium‐Catalyzed Asymmetric Allylic Alkylation Using Chiral ‐Seleno Amides - Braga - 2006 - European Journal of Organic Chemistry - Wiley Online Library
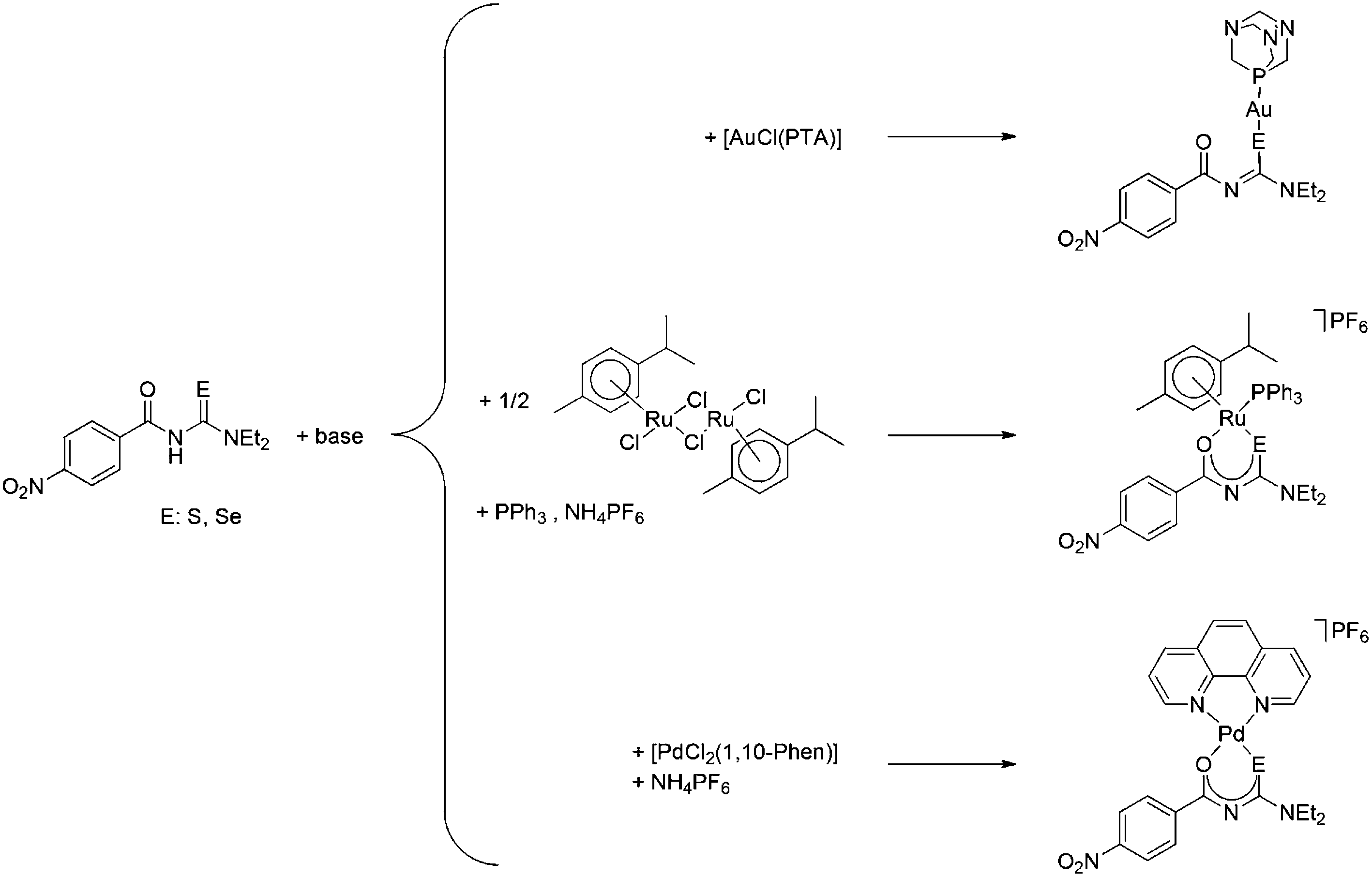
Anti-tumour active gold(i), palladium(ii) and ruthenium(ii) complexes with thio- and selenoureato ligands: a comparative study - Dalton Transactions (RSC Publishing)

PDF) Novel seleno-hydantoin palladium(II) complex - antimigratory, cytotoxic and prooxidative potential on human colon HCT-116 and breast MDA-MB-231 cancer cells

PDF) Synthesis and Properties of Seleno-analog MK-organic Dye for Photovoltaic Cells Prepared by C–H Functionalization Reactions of Selenophene Derivatives
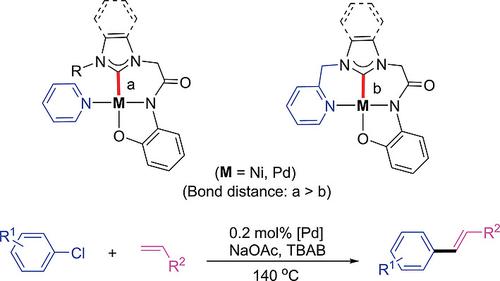
Multidentate N‐Heterocyclic Carbene Complexes of Nickel and Palladium: Structural Analysis and Catalytic Application in Mizoroki–Heck Coupling Reaction - Eur. J. Inorg. Chem. - X-MOL








