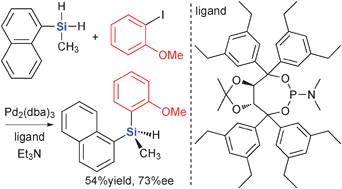
Synthesis of optically active tertiary silanes via Pd-catalyzed enantioselective arylation of secondary silanes - Chemical Communications (RSC Publishing)

Preparation of allyl and vinyl silanes by the palladium-catalyzed silylation of terminal olefins: a silyl-Heck reaction. - Abstract - Europe PMC

Synthesis of α-tertiary allylsilanes by palladium-catalyzed hydrosilylation of 1,1-disubstituted allenes - ScienceDirect
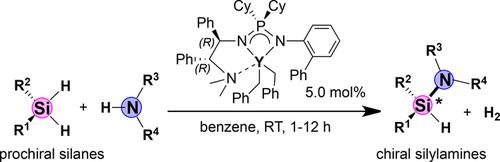
Yttrium–Benzyl Complexes Bearing Chiral Iminophosphonamide Ligands: Synthesis and Application in Catalytic Asymmetric Amine‐Silane Dehydrocoupling Reactions - Adv. Synth. Catal. - X-MOL
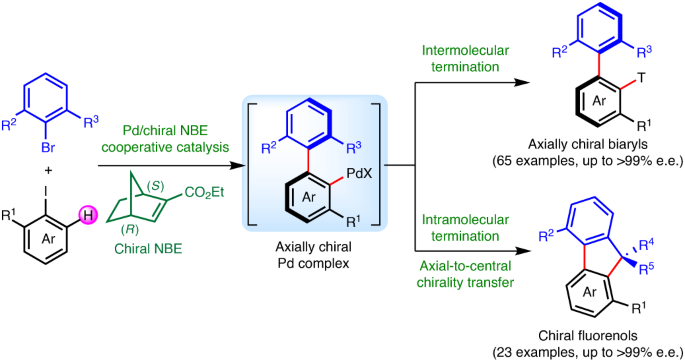
Construction of axial chirality via palladium/chiral norbornene cooperative catalysis | Nature Catalysis

PDF) Si–H Bond Activation of a Primary Silane with a Pt(0) Complex: Synthesis and Structures of Mononuclear (Hydrido)(dihydrosilyl) Platinum(II) Complexes
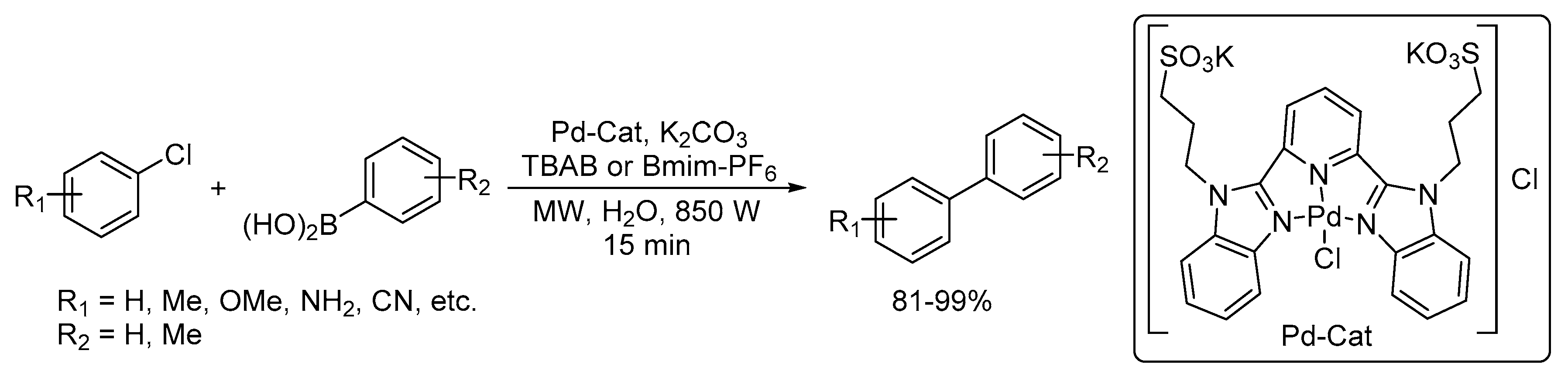
Catalysts | Free Full-Text | Microwave-Assisted Palladium-Catalyzed Cross-Coupling Reactions: Generation of Carbon–Carbon Bond | HTML
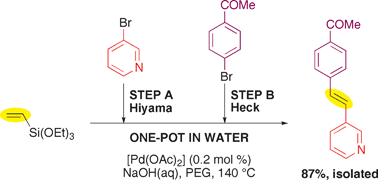
Consecutive palladium-catalyzed Hiyama–Heck reactions in aqueous media under ligand-free conditions - Chemical Communications (RSC Publishing)
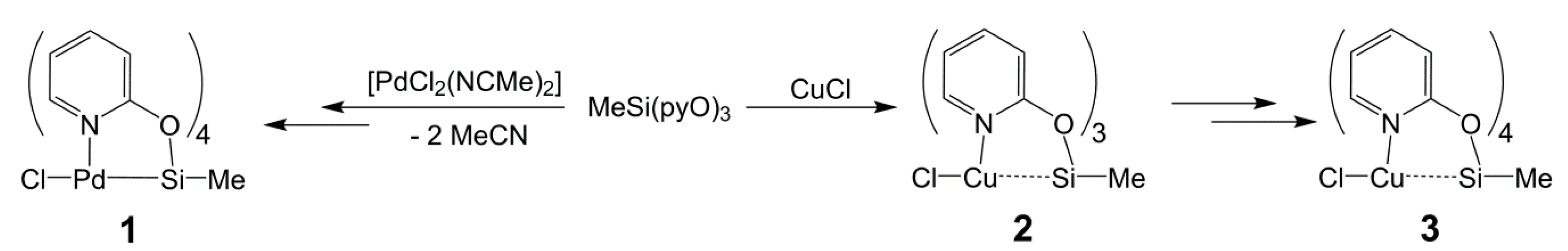
Inorganics | Free Full-Text | (2-Pyridyloxy)silanes as Ligands in Transition Metal Coordination Chemistry
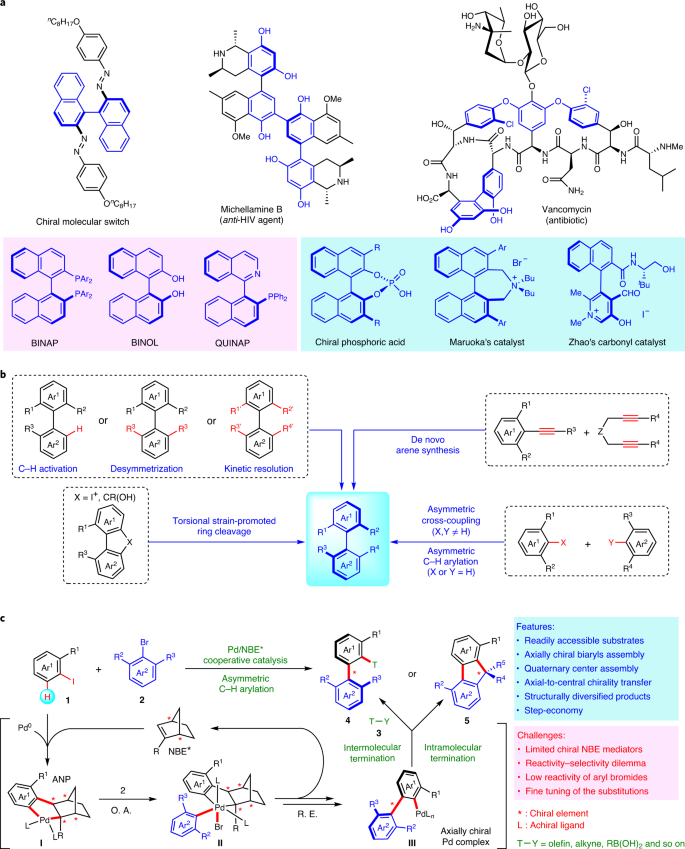
Construction of axial chirality via palladium/chiral norbornene cooperative catalysis | Nature Catalysis

Tailored Palladium Catalysts for Selective Synthesis of Conjugated Enynes by Monocarbonylation of 1,3‐Diynes - Liu - 2020 - Angewandte Chemie - Wiley Online Library
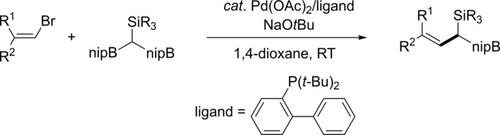
Chemoselective Palladium‐Catalyzed Suzuki‐Miyaura Cross‐Coupling of (Diborylmethyl)silanes with Alkenyl Bromides - Asian J. Org. Chem. - X-MOL
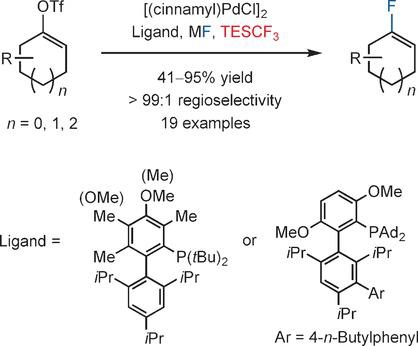
Palladium‐Catalyzed Fluorination of Cyclic Vinyl Triflates: Effect of TESCF3 as an Additive - Angew. Chem. Int. Ed. - X-MOL

Methoxycarbonylation of olefins catalysed by homogeneous palladium(II) complexes of (phenoxy)imine ligands bearing alkoxy silane groups - ScienceDirect

Synthesis of α-tertiary allylsilanes by palladium-catalyzed hydrosilylation of 1,1-disubstituted allenes - ScienceDirect





